XACS Introduction III: MLatom
Published Time: 2022-08-12 17:12:30
We have recently released the Xiamen Atomistic Computing Suite (XACS), which consists of three core software packages:
• XMVB – A Valence Bond Theory Based Quantum Chemistry Program
• XEDA – A General and Multipurpose Energy Decomposition Analysis Program
• MLatom – A Package for Atomistic Simulations with Machine Learning
In our previous post, we introduced XMVB and XEDA packages and will now introduce the third package in the suit – MLatom.
Machine learning (ML) is increasingly used for atomistic simulations, e.g, for representing potential energies, dynamics, spectra prediction, analyses of simulation results, design of new materials, and improving quantum chemical methods. Usually, such application of ML requires programming skills. This and the deluge of ML approaches hinders the routine use of ML in computational chemistry.
MLatom@XACS breaks the barriers to starting using ML in computational chemistry by providing black-box, user-friendly integrative platform for a wide variety of state-of-the-art atomistic machine learning simulations which can be used by both beginners and experts. Programming skills are not required for many common computational chemistry simulations such as geometry optimizations, calculation of thermochemical properties, spectra etc. XACS cloud computing allows users to perform MLatom simulations without any installations just using a web browser (no payment is currently collected!), while advanced users can download and install open-source, free MLatom packages on their computers. MLatom has been developed by Prof. Pavlo Dral, his group, and external contributors for almost 10 years (since 10 September 2013). We provide user support via many channels convenient for users such as MLatom website (http://MLatom.com) and the comments section of its pages, its forum, WeChat official account, Slack, and email. New contributors to MLatom source code are also welcome, please contact us!
One of the highlights of MLatom is general-purpose machine learning methods such as AIQM1 (semi-empirical + machine learning, Nat. Commun. 2021, 12, 7022) and ANI-1ccx (pure machine learning) which allow for CCSD(T)/CBS-quality calculations thousand times faster than B3LYP/6-31G* (no training required!). In addition, we continuously release novel methods we develop, test, and publish, so that they are immediately available for use by our community.
MLatom's capabilities are quite broad ranging from various data to ML tasks, whose implementations are constantly expanding and improving. Here we just list some major features:
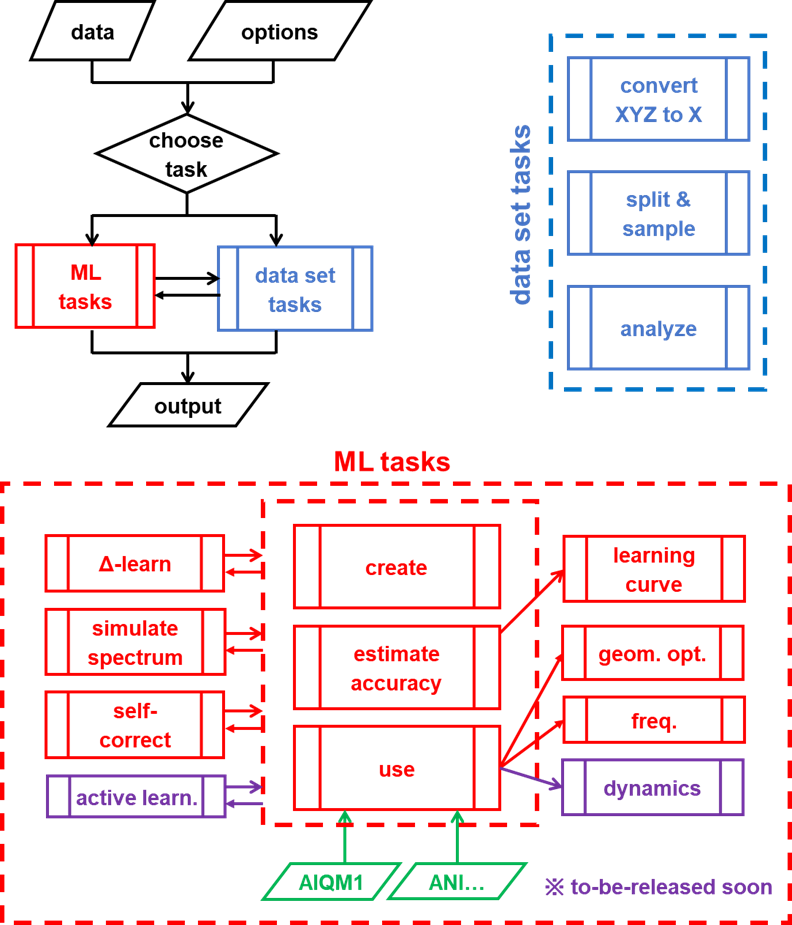
We cannot demonstrate all the features of MLatom in a single post, thus, here we will show just a single example and we will continue preparing posts about other features. More tutorials and manuals you can find on http://MLatom.com. The example below will be shown for the MLatom@XACS version based on XACS cloud computing.
1. Starting off
With XACS cloud computing, users do not need to download or install software packages. You can simply visit the XACS official website (http://xacs.xmu.edu.cn), register for free or login to your existing account, submit calculations, track the progress and retrieve the calculation results.
2. Example
Take the thermochemical calculations of vinylacetylene as an example, which is available on the XACS website (https://xacs.xmu.edu.cn/records?attribution_program=MLatom). Here are the input and auxiliary files:
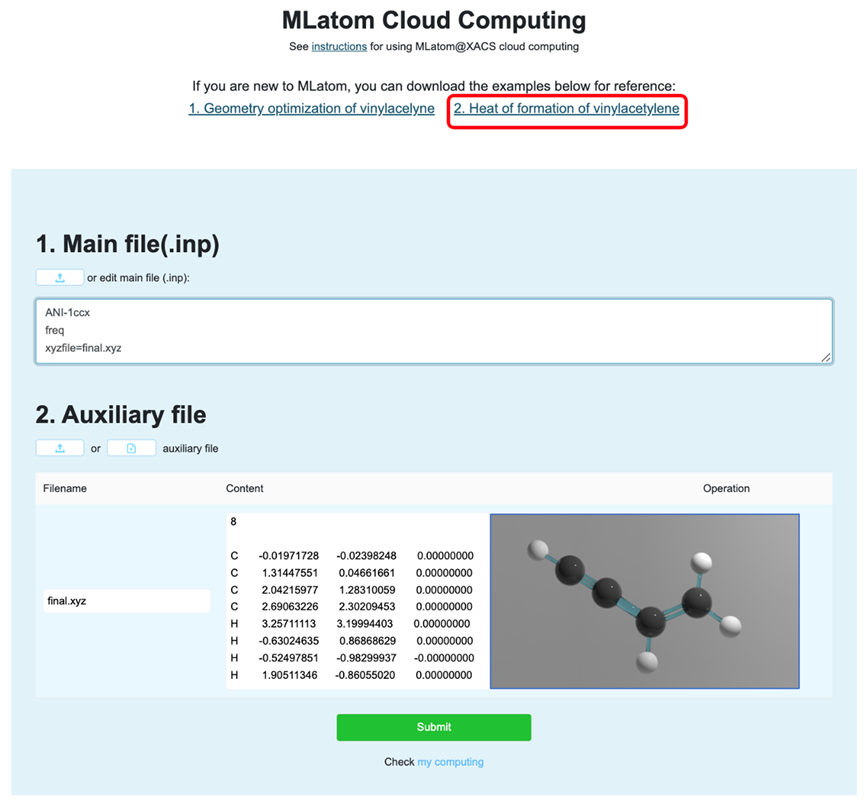
Input file specifies the type of MLatom calculations, auxiliary files are required as usually ML deals with big data sets. In our example, MLatom input file requests frequency calculations with freq keyword and using ANI-1ccx method (provided by python module TorchANI). Auxiliary file contains geometry of vinylacelyne optimized at ANI-1ccx.
Visit the MLatom cloud computing page and click Submit to start the MLatom calculations. It should take couple of seconds after which you can download result in a zipped folder containing log file with MLatom output, which contains a summary of thermochemical calculations like this:
Congratulations, you have just calculated the heat of formation of vinylacetylene in seconds and with accuracy comparable to much slower high-level quantum chemical methods! In fact, ANI-1ccx heat of formation (68.5 kcal/mol) is very close to one of the experimental values (70.4 kcal/mol) and helped us (J. Phys. Chem. Lett. 2022, 3479) to revise a previously used experimental value (73 kcal/mol).
Welcome to visit the XACS website for more help. If you have any questions, please contact us via xmvb@xmu.edu.cn.
XACS homepage:http://xacs.xmu.edu.cn
MLatom manual:http://mlatom.com/manual/
MLatom tutorial:http://mlatom.com/tutorial/
MLatom@XACS instructions: http://mlatom.com/mlatom-at-xacs-cloud-computing/